Metrion was an early and active participant in the Comprehensive In vitro Proarrhythmia Assay (CiPA) ion channel high-throughput screening (HTS) sub-team, playing a key role in advancing the initiative. In addition to our involvement with CiPA, we were also an integral member of the Health and Environmental Sciences Institute (HESI) cardiac committee, where we collaborated closely with HESI and other stakeholders in the field to enhance the accuracy and reliability of cardiac safety assessments.
Our expertise was valuable in helping to reduce data variability across multiple screening sites, ensuring that results were consistent and reproducible. Furthermore, we provided crucial validation data to support the development of predictive in silico models, which have become an essential tool in risk assessment for drug-induced arrhythmias.
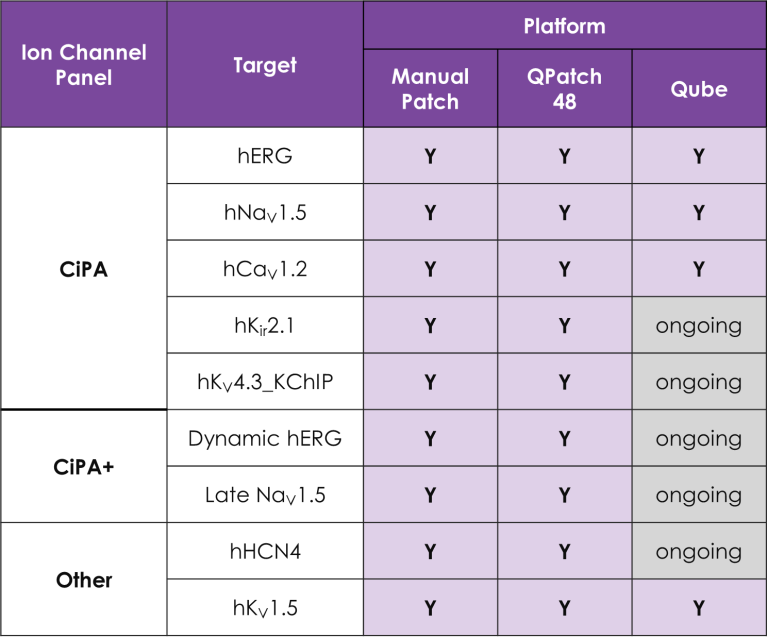
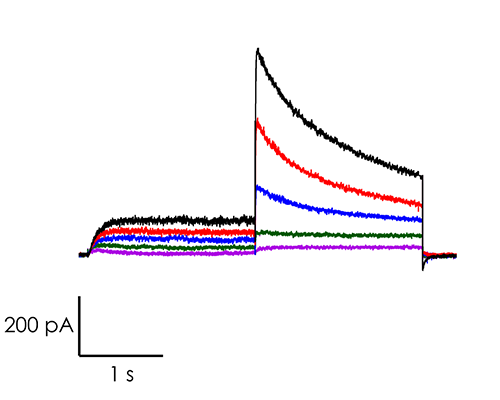
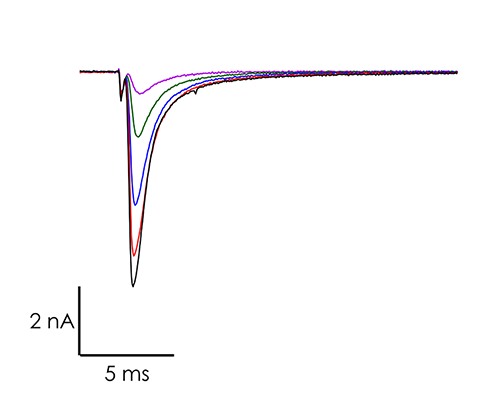
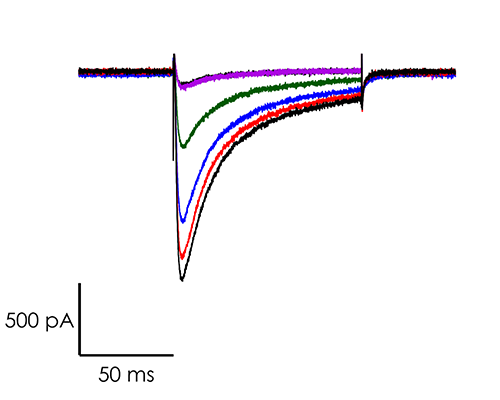
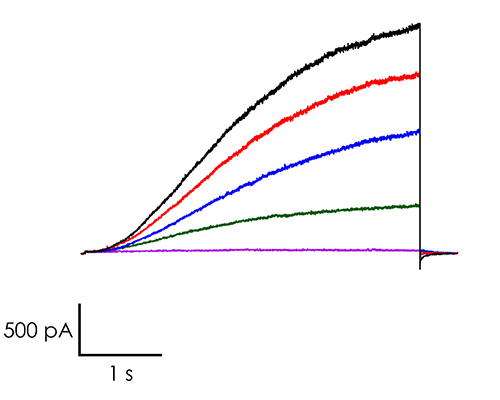
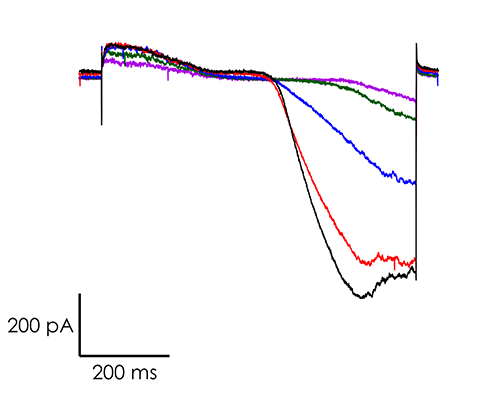
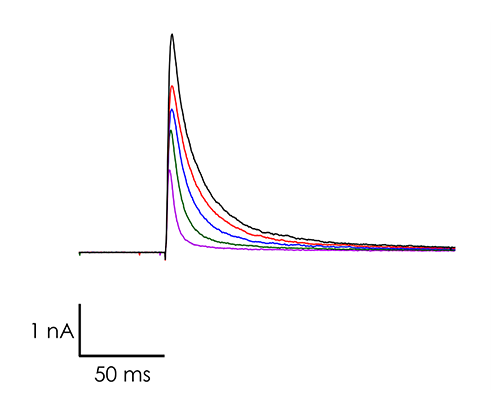
A portfolio of highly validated assays targeting a broad range of human ventricular ion channels, including those that form the full CiPA panel are developed and maintained by Metrion: hERG (human Ether-à-go-go-Related Gene), peak and late Nav1.5 (sodium channel), Cav1.2 (L-type calcium channel), KCNQ1/KCNE1 (potassium channels), Kir2.1 (inward rectifier potassium channel), and Kv4.3 (a component of the delayed rectifier potassium channel) as outlined in Table 1. In addition, we have pioneered the development of a robust dynamic hERG assay, which has been leveraged to provide critical data that contributes to improving the accuracy of the Food and Drug Administration’s (FDA) in silico qNet model, a tool used for predicting cardiac risk in drug development. Beyond the CiPA panel, Metrion offers comprehensive screening services for additional ion channels such as HCN4 and Kv1.5, which play pivotal roles in regulating human heart rate and atrial repolarisation, respectively. Our ability to offer these screening services makes us a valuable partner in advancing cardiac safety and improving the overall efficiency of the drug development process.
Table 1. Assays for a wide range of different human cardiac ion channels.