By accurately defining the drug exposure levels that affect QRS duration, researchers can establish safety margins, prioritise lower-risk compounds, and reduce the chance of late-stage failures due to cardiac toxicity
Metrion has extensive ion channel drug discovery experience of indications such as pain, neurodegeneration, psychiatry and epilepsy.
As our specialist team of neuroscientists are experts in their field we can advance your project to the next milestone, as well as help your biology team generate cell lines for your targets.
We have extensive experience in electrophysiology and ion channel screening assays, and translational assays, delivering high-quality recordings from relevant tissues expressing your target of interest.
We offer high-throughput automated patch clamp services, fluorescence, MEA and manual patch (intracellular/organelle recordings).
Our panel of models allow for more accurate prediction of human responses, reduce the risk of late-stage failures and provide a thorough understanding of drug mechanisms.
Ion channel cell lines for both central and peripheral nervous system targets
Primary neuronal cultures, such as dissociated cortical, trigeminal or dorsal root ganglion (DRG) neurons from rodents
CNS drug discovery: Increase the likelihood of successful therapeutic outcomes
Human iPSC-derived neurons

Figure 1. Electrophysiological recordings of primary neurons. A. Rodent dorsal root ganglion (DRG) neuron preparation for manual patch-clamp studies and a bright-field image of dissociated cells seeded on a coverslip. B. DRG action potential responses to low and high prolonged current injections.
A range of assay methodologies allows a greater understanding of the mechanism and impact of ion channel modulation on neuronal firing and network activity. Examples of the assays we offer include:
Manual and automated patch-clamp
Multi-electrode array (MEA) recordings
Translational assays: central neuronal firing, peripheral neuronal firing, native ion channels
Our neuroscience assays are used to:
Investigate the mechanism of action for pre-clinical compounds in neuroscience discovery programmes
Confirm compound effects against specific ion channels in native neuronal backgrounds
Address the neurotoxicity of compounds and assess the effects of compounds on a range of excitability endpoints
Our neuroscience experts can evaluate potency and mechanism of action of compounds using heterologous cell lines expressing human or rodent neuronal ion channels (including challenging to develop cell reagents such as NaV1.8 and NaV1.9 lines). Effects of compounds on neuronal excitability can be assessed using human iPSC-derived neurons and primary rodent neurons.
Get insight into safety profiling of lead compounds and IND candidates, either as part of an existing screening cascade or because in vivo testing revealed a neurological signal. We have developed and validated an industry-standard rat cortical CNS neuron assay that can reliably detect the seizure-inducing liability of a wide range of reference compounds (as well as serve as a model to profile anti-epileptic drugs). We also have deep expertise with rodent dorsal root ganglion (DRG) sensory neurons which can be used to assess peripheral neurotoxicity.
Studying ion channels on the lysosomal membrane is essential for understanding lysosomal function, cellular homeostasis, and the development of various neurological diseases, such as Parkinson's disease. This research has the potential to uncover new therapeutic targets and improve our knowledge of fundamental cellular processes.
Read more about how we perform the lysosomal patch clamp technique to study ion channels on the lysosomal membrane, and watch this video to see it in action.
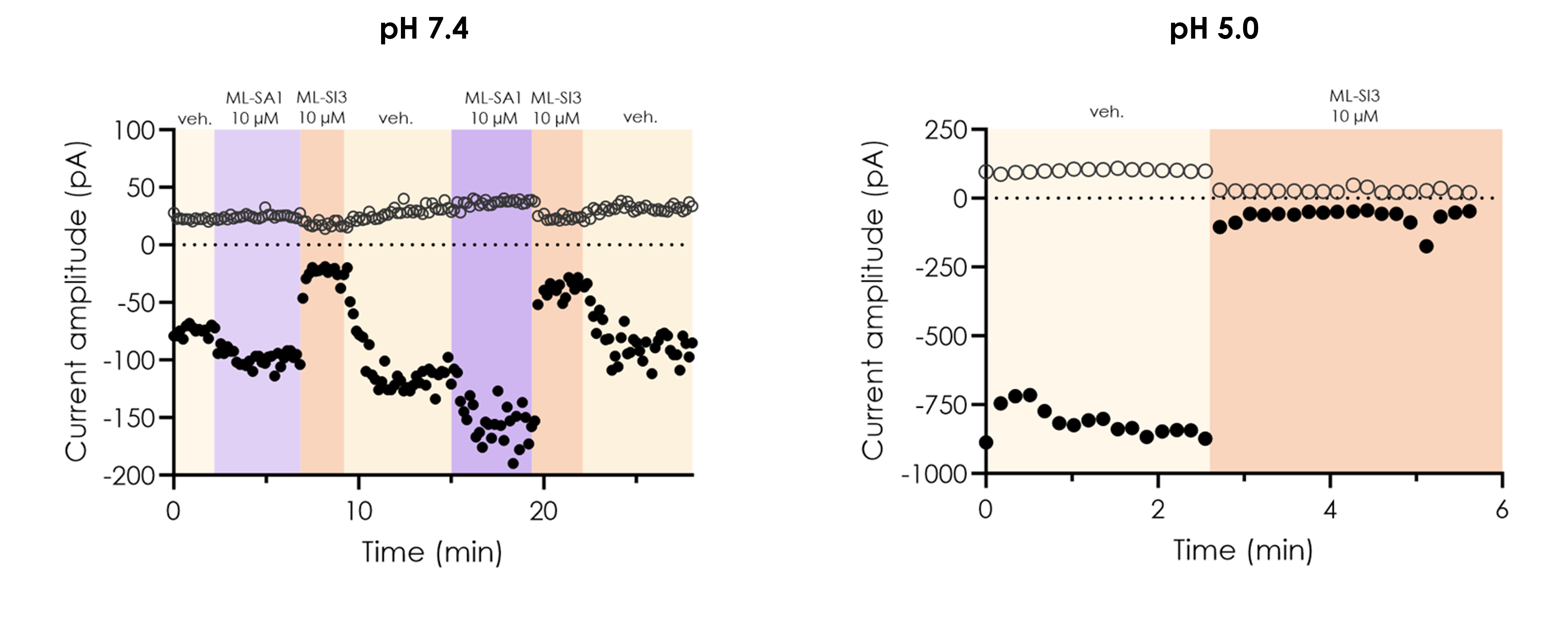
Figure 2. Pharmacological inhibition of wild-type TRPML1 currents recorded by manual patch-clamp.
Eliana is a two-year-old from Canada with a de novo mutation (V434L) in her KCNC1 gene which encodes for the Kv3.1 channel in central nervous system neurons such as cerebellar neurons and GABAergic interneurons. The mutation manifests as a variety of neurological disorders which can include myoclonic epilepsy and ataxia, developmental epileptic encephalopathy (DEE), or hypotonia, depending on the specific variant.
The KCNC1 Foundation collaborated with Perlara, who approached Metrion Biosciences, where manual and automated (Qube) patch-clamp techniques and Fluorescent Imaging Plate Reader (FLIPR) high-throughput screens (HTS) against the mutant channel were performed to identify hit compounds.
By accurately defining the drug exposure levels that affect QRS duration, researchers can establish safety margins, prioritise lower-risk compounds, and reduce the chance of late-stage failures due to cardiac toxicity
By accurately defining the drug exposure levels that affect QRS duration, researchers can establish safety margins, prioritise lower-risk compounds, and reduce the chance of late-stage failures due to cardiac toxicity