The key question is no longer whether to adopt NAMs, but how to implement them with confidence while continuing to meet evolving regulatory expectations and protecting programme value.
Our expert electrophysiologists and cell biologists help you identify and de-risk proarrhythmic and cardiotoxic liabilities with high-quality data, rapid turnaround times and scientifically robust interpretation. Comprehensive cardiac safety assays are offered for early discovery, lead optimisation and IND-enabling studies, including GLP hERG testing, CiPA ion channel profiling and clinically translatable hiPSC cardiomyocyte assays.
Our cardiac safety team is led by experts who have exceptional ion channel electrophysiology and drug discovery expertise, complemented by a team of experienced cell biologists who create novel cell lines, to offer:
Ion channels play a central role in cardiac electrophysiology and are key targets in preclinical cardiac safety assessment. Our ion channel screening services provide high-quality electrophysiology data to evaluate proarrhythmic liability and support informed decision-making during drug discovery.
Our experienced scientists deliver detailed characterisation of compound effects using validated assay platforms and flexible screening strategies tailored to your project requirements.
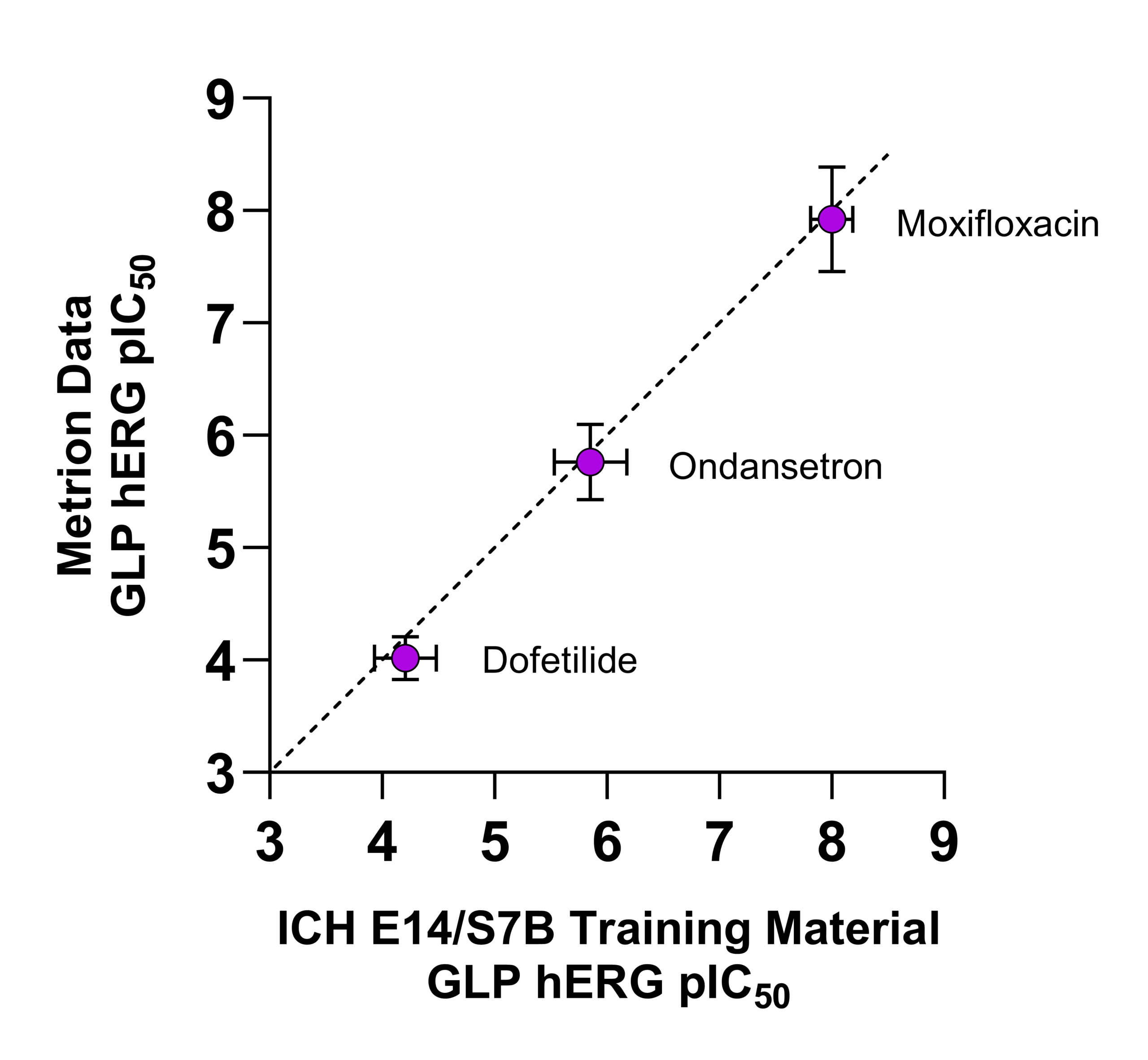
Right: Comparison of GLP hERG IC50 values from Metrion versus published ICH E14/S7B training material values.


Our cardiac safety team combines exceptional ion channel electrophysiology expertise with extensive drug discovery experience.
We deliver scientifically rigorous data generated using validated assays and robust experimental design. Reports are not just a summary but a strategic tool to support your decision-making and move your research forward with confidence.

We tailor screening strategies to your project stage, timelines and budget requirements.
Our integrated ion channel and hiPSC cardiomyocyte assays provide clinically relevant cardiac safety assessment.
Fast study execution and expert interpretation help accelerate decision-making during drug discovery.
Rapidly identify potential cardiac liabilities during hit identification and lead optimisation using high-throughput ion channel and hiPSC cardiomyocyte assays. These approaches support early decision-making by enabling efficient SAR screening, early hERG assessment, and prioritisation of lead compounds, while helping to identify potential QT liability risks before significant development investment.
Recommended assays for this stage include hERG screening, core ion channel profiling, and hiPSC voltage dye assays.
As compounds advance, deeper insight into cardiac electrophysiology liabilities becomes essential. Expanded ion channel panels and translational phenotypic models provide mechanistic understanding of compound effects, supporting multi-ion-channel assessment, mechanistic electrophysiology studies, CiPA profiling, and prediction of QRS liability.
Recommended assays include expanded CiPA panels, hiPSC current clamp profiling, and advanced ion channel profiling.
For later-stage development and regulatory submission, GLP-compliant cardiac safety studies provide robust data aligned with current ICH S7B guidance. These studies support IND submissions, regulatory risk assessment, and confirmatory cardiac safety evaluation through validated testing strategies and expert interpretation.
Recommended assays include GLP hERG testing and translational hiPSC cardiomyocyte assays.
Cardiac safety screening evaluates whether a compound may interfere with cardiac electrophysiology or cause cardiotoxicity. Common approaches include ion channel assays, hERG testing, CiPA profiling and hiPSC cardiomyocyte models.
The hERG assay assesses whether compounds inhibit the hERG potassium channel, which is associated with QT prolongation and increased risk of ventricular arrhythmias.
hERG screening evaluates a single cardiac ion channel, while CiPA screening assesses multiple ion channels and incorporates mechanistic approaches to improve prediction of proarrhythmic risk.
hiPSC cardiomyocyte assays use human stem cell-derived cardiac cells to assess electrophysiological and cardiotoxic effects in a clinically relevant cellular model.
GLP hERG studies are commonly included in regulatory cardiac safety packages to support IND submissions and demonstrate compliance with current ICH S7B guidance.
The key question is no longer whether to adopt NAMs, but how to implement them with confidence while continuing to meet evolving regulatory expectations and protecting programme value.
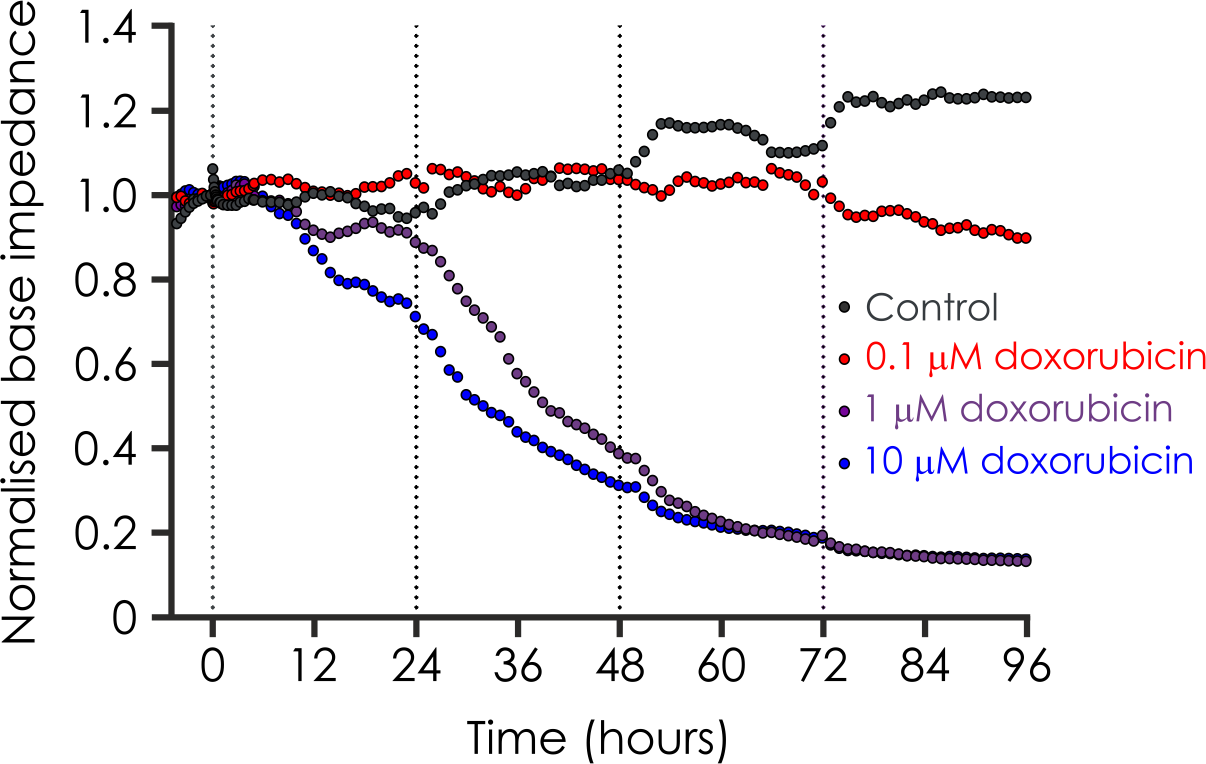
Optical voltage imaging of human iPSC-derived cardiomyocytes was used to assess electrophysiological effects of compounds beyond hERG inhibition. Action potential waveform analysis revealed compound-specific and concentration-dependent changes, enabling mechanistic differentiation of multichannel activity and demonstrating a human-relevant approach for translational cardiac safety assessment.